- Home
- Industry
- Products
- Services
- Cloud Platform
Enquire Now

Enquire Now

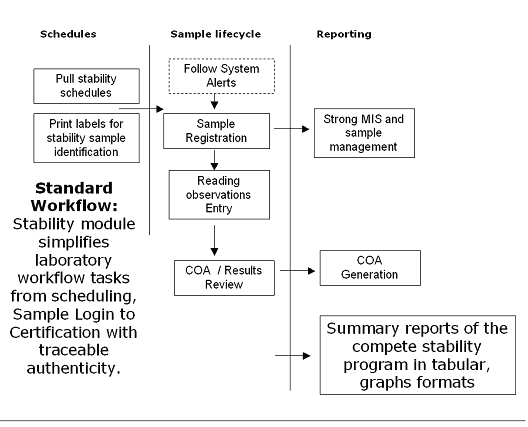
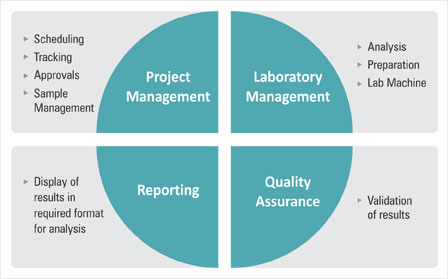
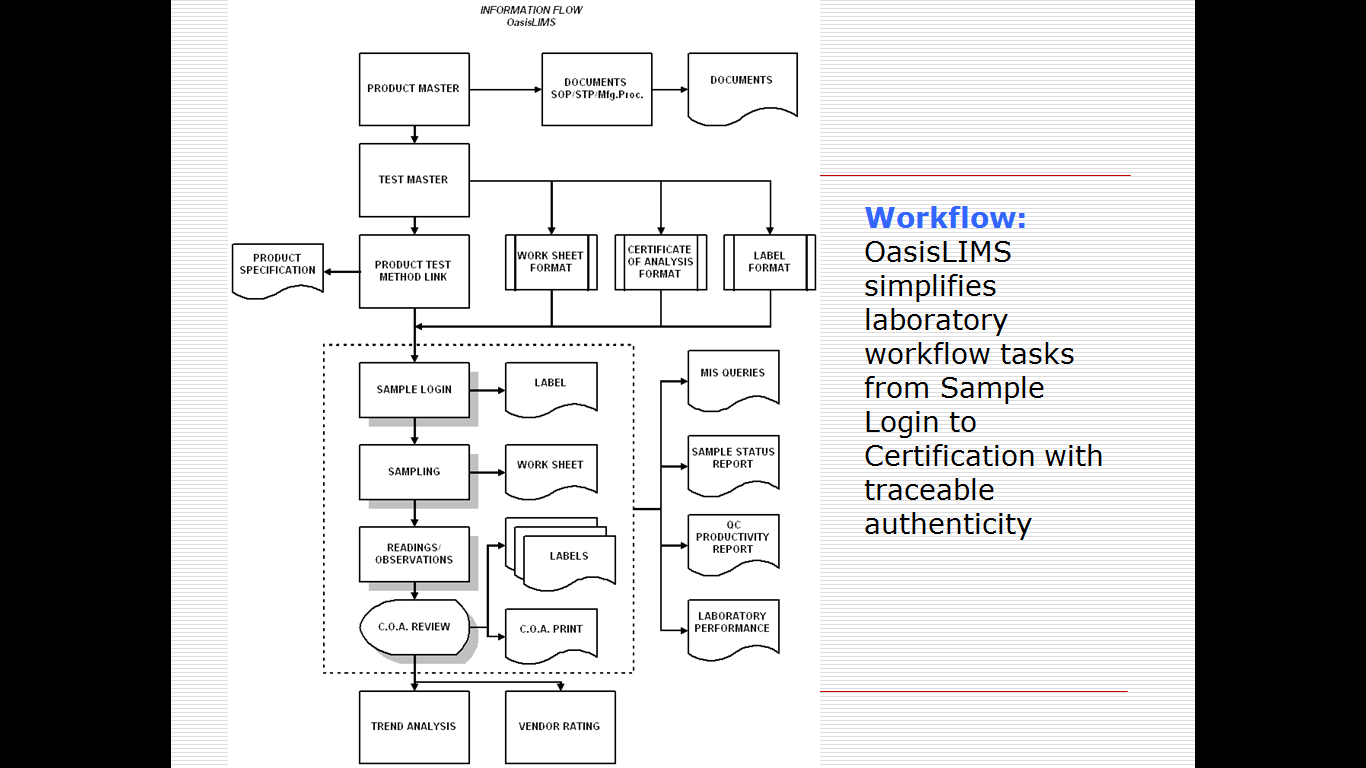
OasisLIMS Professional is a comprehensive web based and client / Server based standard laboratory information management software, it is suitable small and mid size laboratories for a wide range of laboratory applications. OasisLIMS Professional’s native functionality allows you to effectively manage the flow of samples and associated data to improve lab efficiency; it helps standardize workflows, tests and procedures, while providing accurate controls of the process. Automatic on-line calculations are integrated into the LIMS to record collection of test data and eliminate error, lab resource allocation module ensures that instruments used in analysis are properly calibrated and operated by trained/qualified staff only. It also ensures chemicals; standards and reagents used in performing analysis are suitable for their intended use.
OasisLIMS key functions include revision controls of masters incline organizations to use a LIMS and ‘go paperless’ to meet best practice or compliance requirements, for pharma or other regulatory major organizations OasisLIMS professional may also be configured to incorporate audit trails to capture changes to results, test procedures, instruments and chemicals used during analysis. Implementing OasisLIMS professional saves valuable man-hours of technical staff, saves time spent in writing and checking in individual reports. The time saved – utilized for other important / productive tasks in the lab. The main purpose of a OasisLIMS is to improve lab efficiency and accuracy by reducing manual operations, it will performs a range of core functions that includes
Sample Management / Sampling / Worksheets / Workflow management / Test data record keeping / Automatic test method calculations / Inventory management / Instruments management / Reporting / Administration and control / Flexible Reporting / Trending and Analytical Quality Control / Standard Operating Procedures Maintenance / Instruments Management / Inventory Management / Auditing and Security / Simple User Interface
Facility to import SOPs from stored files (maintained under standard word processor like MS Word, ASCII Text, etc.)