- Home
- Industry
- Products
- Services
- Cloud Platform
Enquire Now

Enquire Now

All Oasis network system products incorporate functions as per regulatory guidelines and computerized validation functions required by GXP. Oasis provides full documentation including IQ/OQ, Certificate of compliance, and inspection test result reports based on certified system. Oasis total solution for Pic/s, GMP, FDA 21 CFR Part 11 and computerized validation – establishes proof for reliability and security built in the system. Systematic validation support for creating system operation and management procedures required for FDA compliance by providing ready to use DQ / OQ / OQ computer validation templates.
Oasis offers validation and documentation assistance to customers on the basis system configuration in accordance preparation of documents in such a way that relevant and applicable parameters of V-model life cycle are followed appropriately as a standard.
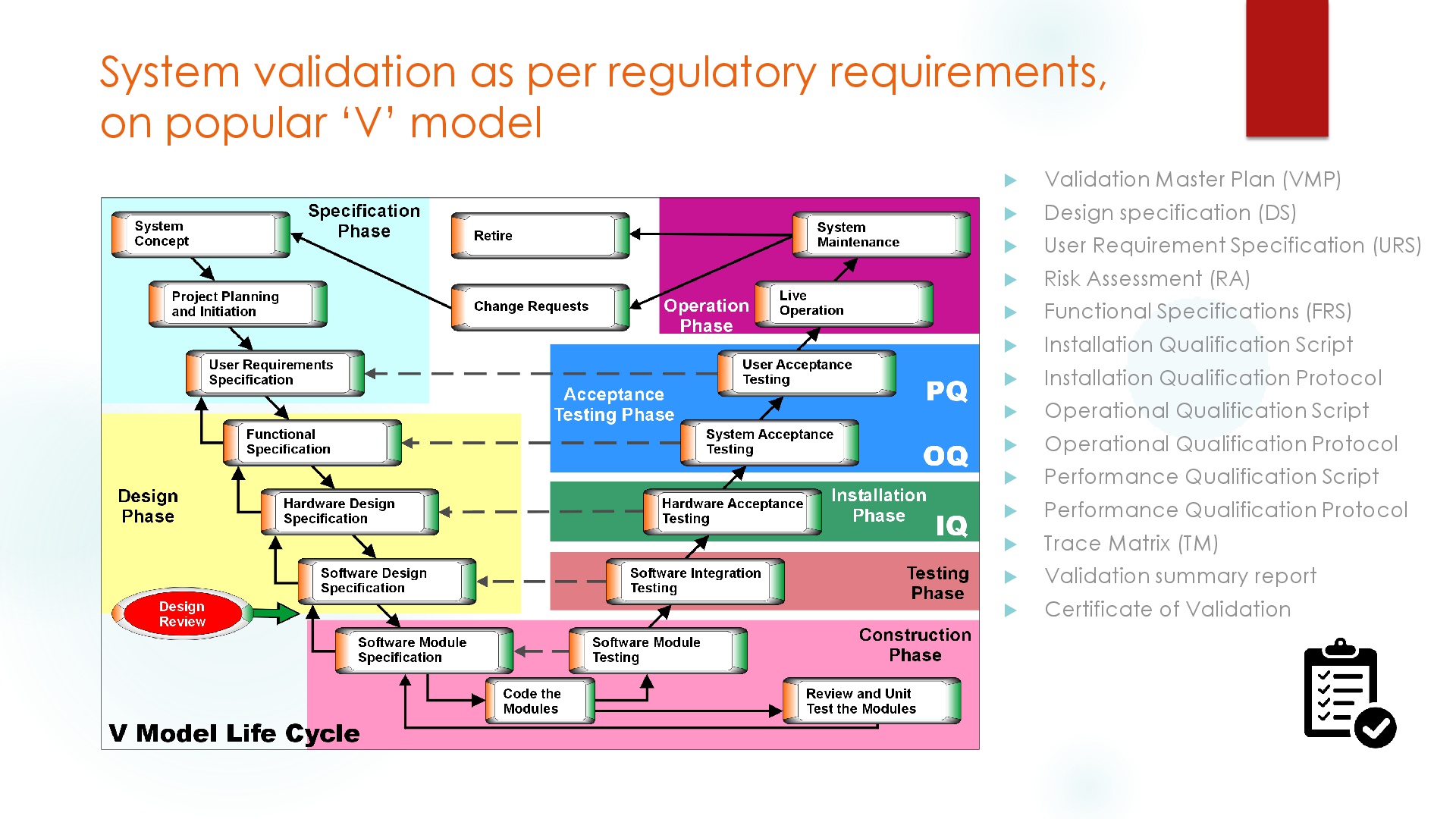
Project plan including training plan / training logs, System concept including risk analysis, Hardware design / requirement specifications
User requirement specification, Functional specifications, Traceability matrix (against URS, FS)
Software installation acceptance testing including network qualification Traceability matrix (against IQ)
Manuals / SOP’s
System / user acceptance testing
Test protocol results
Problem resolution forms
Operational / performance qualification report
Traceability matrix (against OQ/PQ)
Validation report and summary, Certificates