- Home
- Industry
- Products
- Services
- Cloud Platform
Enquire Now

Enquire Now

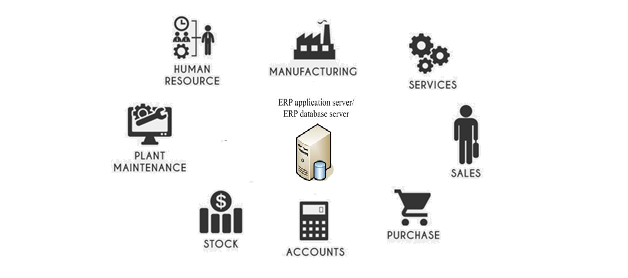
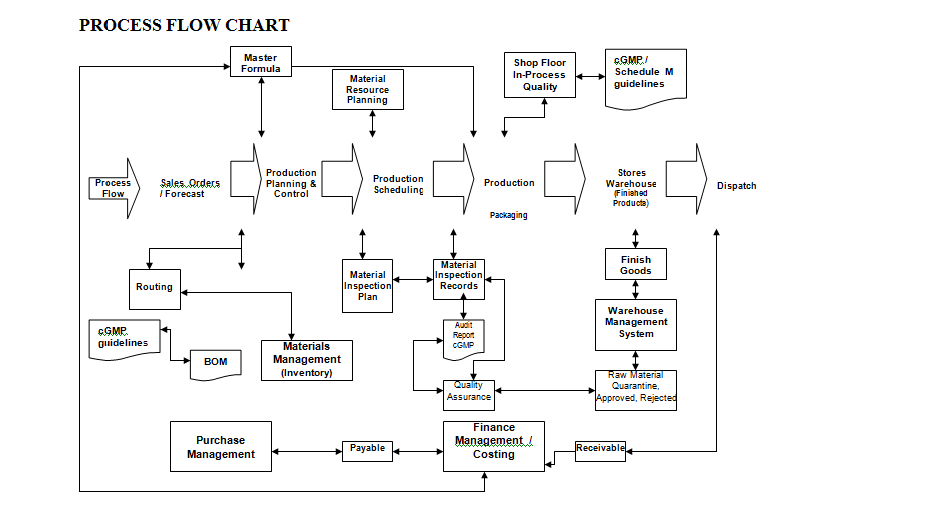
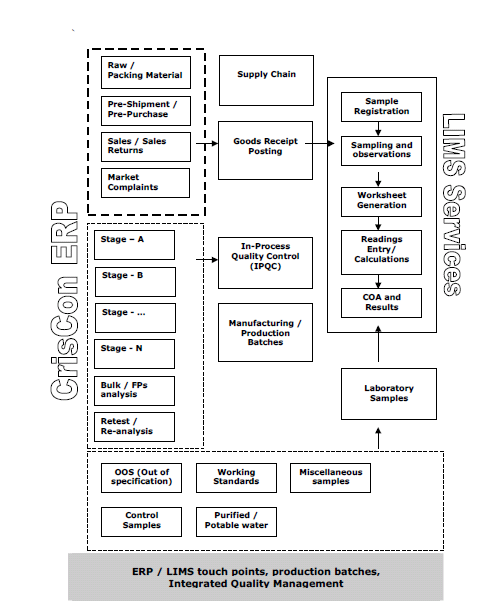
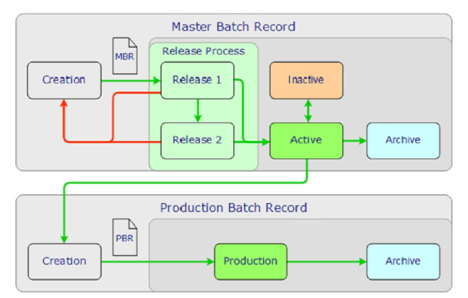
Calibration scheduling, calibration results recording
Scheduling of the preventive maintenance and capturing the preventive maintenance details
Problem reporting and capturing of the service details
Management of Annual Maintenance Contracts
Engineering Inventory Management
Management of Work orders
Engineering Inventory Management
Operations/usage logs
Cleaning Logs
System keeps track of instrument history maintenance records
• Test library for laboratory tests
• Test profiles
• Test Details - Test Limits / formats